Everything your pharmacy needs for 2025 DSCSA compliance right here!
The DSCSA regulation has 2 primary components
Verification
- Determine whether trading partners who you do business with are licensed or registered.
- Retain the tracing information (T3) on each product for at least 6 years.
Reporting
- Grant Product Tracing Documentation within 48 hours of a request.
- Report suspicious or illegitimate drug products within 24 hours of discovery.
If you are a pharmacy or a prescription drug distributor, the Drug Supply Chain Security Act (DSCSA) affects your business.
Prescription drug traceability can involve large investments. We are revolutionizing access to traceability technologies and enabling every pharmacy in the country to meet DSCSA requirements in a timely and cost effective manner.
START YOUR JOURNEY NOW
CREATE A FREE ACCOUNT
Meet TrackRX, the fast track to traceability & regulatory compliance
BASIC
FREE-
TrackRX Traceability Portal
-
DSCSA Validation Module
-
Serialization EPCIS / Support
-
Unlimited Transaction
-
Unlimited T3/T2 Manual Creation
-
Unlimited SKUs
-
Six Years Retention
-
1 Location
-
1 Trading Partner
-
1 User creation
-
500 Megs Storage
-
Email Support
MEDIUM
CONTACT FOR PRICING-
TrackRX Traceability Portal
-
DSCSA Validation Module
-
Serialization EPCIS / Support
-
Unlimited Transaction
-
Unlimited T3/T2 Manual Creation
-
Unlimited SKUs
-
Six Years Retention
-
Unlimited Location
-
Unlimited Trading Partner
-
Unlimited User creation
-
Unlimited Storage
-
Email Support / Phone Support
PROFESSIONAL
CONTACT FOR PRICING-
TrackRX Traceability Portal
-
DSCSA Validation Module
-
Serialization EPCIS / Support
-
Unlimited Transaction
-
Unlimited T3/T2 Manual Creation
-
Unlimited SKUs
-
Six Years Retention
-
Unlimited Location
-
Unlimited Trading Partner
-
Unlimited User creation
-
Unlimited Storage
-
Email Support / Phone Support
-
VRS Access
-
RapidRX Mobile Edge
-
Internal System/ERP/WMS Integration
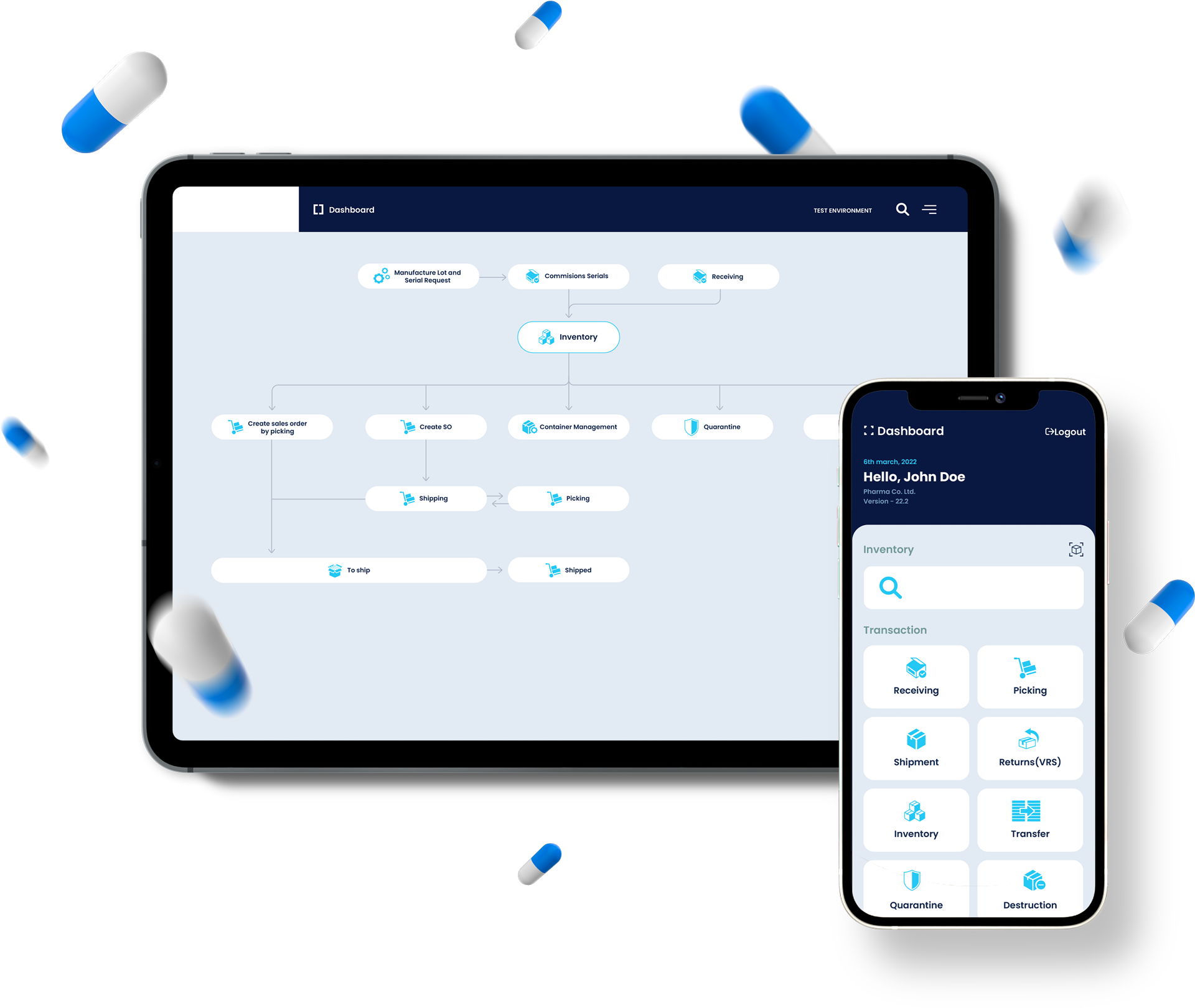
The TrackRX Portal saves weeks of setup and investment to meet any demand or regulatory requirement. Coupled with the automated data collection of the MultiScanner RapidRX app, no additional work is required to collect accurate, human error-free data and provide unmatched insight into your processes.

CLOUD-BASED
Flexibility of connecting to your business anywhere, any time.

EASY TO USE
Screens responsive to any device. Simple and customizable workflow.

SCALABLE
Integrate the portal into your operations without any disruption.

COMPLETELY TRANSPARENT
No hidden fees or later costs.
CREATING A SECURE AND COMPLIANT PHARMA SUPPLY CHAIN
Why choose us?
TrackTraceRX is a pioneer in traceability, with over 15 years experience in the market and leading solutions for the needs of the pharmaceutical industry. Having performed over 100 million transactions worldwide, we are redefining the “Fully Interoperable Solution” for the future of DSCSA 2025.
We have customers using our solutions for over 15 years!
Join our clients





