We Handle ALL Your DSCSA Requirements
- 2025 DSCSA Compliance Solution
- Integrated with over 100,000+ Trading Partner Locations
- DSCSA Software with EPCIS Data Exchange
- VRS & Authorized Trading Partner (ATP) Ready
- Free Trading Partner Onboarding
- Free Audit Support
- Award-Winning Company
The First and Only Solution with Built-in Multi-Scanning and Augmented Reality to
Solve Your DSCSA Requirements, Providing True End-to-End Track & Trace.
Award-Winning Company Trusted by Leading Pharma Companies

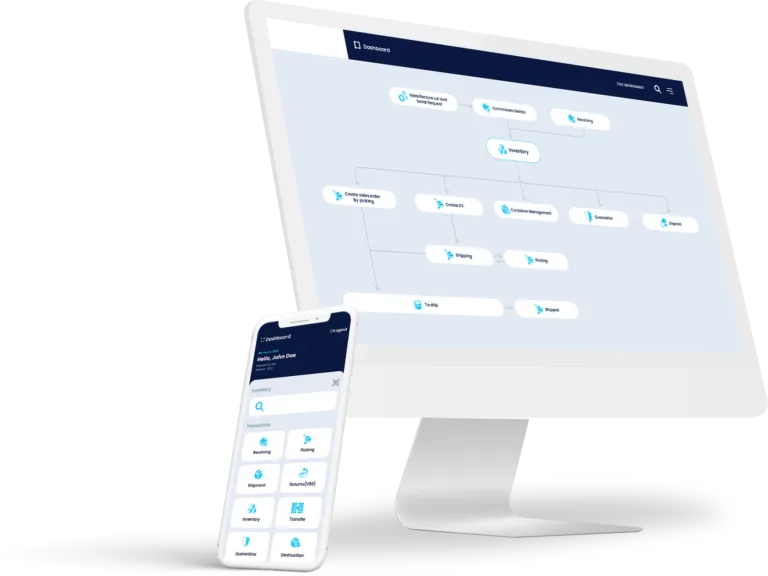
Track & Trace DSCSA Cloud Portal
End-to-End Tracking:
TrackRX monitors the movement of products and drugs throughout the entire supply chain. From manufacturing and packaging to handling, distribution, prescription, and final delivery, ensure complete visibility and control. The best part? we are already Integrated to over 100,000+ partner locations. This means you only need to integrate once to become a part of our extensive network.
Free Trading Partner onboarding:
We are the ONLY solution provider that will contact EVERY one of your trading partners and integrate to receive track & trace data for FREE. So you can focus on what you do best – running your business.
Integrate with your Internal ERP or WMS System:
Easily integrate TrackRX with your existing Warehouse Management Systems (WMS), Enterprise Resource Planning (ERP) systems, and other software. Our API-first approach ensures seamless connectivity and data exchange.

The First and Only DSCSA smart data capture solution
Powerful AR Multi-Scanner: Designed for accuracy and speed in barcode scanning, RapidRX takes track & trace and DSCSA compliance to a whole new level. This technology delivers unparalleled mobility for your operations by utilizing a smartphone or tablet, and it is uniquely integrated with Augmented Reality (AR).
- Mobile software that reduces normal costs with dedicated scanners.
- Scans DSCSA required shipments and automatically flags any issues using augmented reality.
- Real-time Notifications ideal for VRS and Tracing Requests, providing real-time notifications for swift action.
- High efficiency, resulting in more productivity and profits.

ERPRX Connector – Connect your Systems to TrackRX & RapidRX
Imagine your internal systems, such as an ERP, fully connected to a track & trace system and a mobile edge multi-scanner. With the ERPRX Connector, this vision becomes a reality.
- Easily Integrate with the RapidRX & TrackRX.
- Enhance your systems with the powerful features of the RapidRX Multi-Scan and Augmented Reality capabilities.
- Add Track & Trace to your products with TrackRX.
- Combine ERP, Track & Trace and Mobile Scanning.
- The ERPRX Connector is a database endpoint connector to easily allow different systems the ability to exchange data.
Track & Trace Solutions for Multiple Industries
We offer innovative and collaborative traceability solutions, ensuring seamless integration and visibility across various industries
Automotive industry
Automotive industry
Learn morePharmaceutical industry
Pharmaceutical industry
Learn moreAgrobusiness
Agrobusiness
Learn moreCosmetics industry
Cosmetics industry
Coming soonPharmacies, wholesalers, or manufacturers, the FDA’s exemptions give you additional time to get your DSCSA systems in order.
Our comprehensive TrackRX Portal is your key to navigating this stabilization period with ease, ensuring your operations align with DSCSA mandates without interruption.
We have customers using our solutions for over 15 years!
Join our clients




