
5 Strategies to Combat and Reduce Counterfeit Drugs in the Supply Chain
Read 5 strategies to help safeguard the pharmaceutical supply chain from counterfeit drugs.

Read 5 strategies to help safeguard the pharmaceutical supply chain from counterfeit drugs.
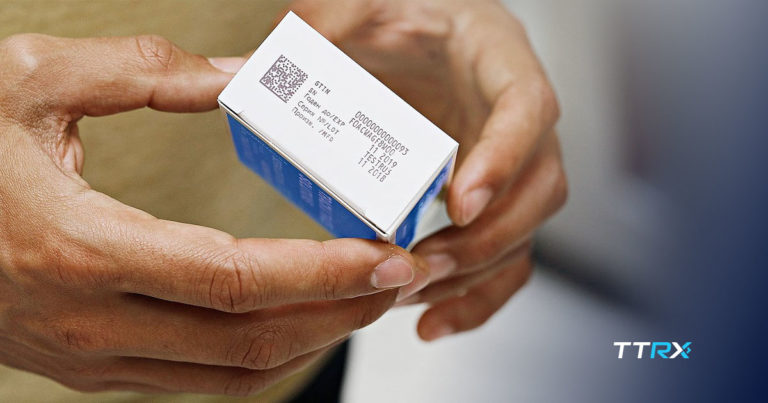
Discover the importance of the GS1 EPCIS standard language in the fight against counterfeit drugs in healthcare and learn why you should prepare now.

The November 27, 2023 deadline is fast approaching for pharmaceutical companies to have full unit-level traceability in compliance with the DSCSA. Read more

The US Food and Drug Administration (FDA) has outlined DSCSA implementation plans in four guidances. Find out more!

Pharmaceutical serialization and traceability is now a requirement with looming deadlines. It’s time to think about compliance. Get ready now!

Find out what the pharmaceutical packaging serialization requirements are and how to turn compliance into a competitive advantage for your company!

Pharmaceutical DSCSA guidance made easy. How does the DSCSA affect consumers?

DSCSA Q/A With Dirk Rodgers from RxTrace
DSCSA – Standard Operating Procedure for Product Tracing
Preparing for Serialization – Focal Points
Shaping the future of traceability and serialization since 2007